 
The cookie is used to store the user consent for the cookies in the category "Other. This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. 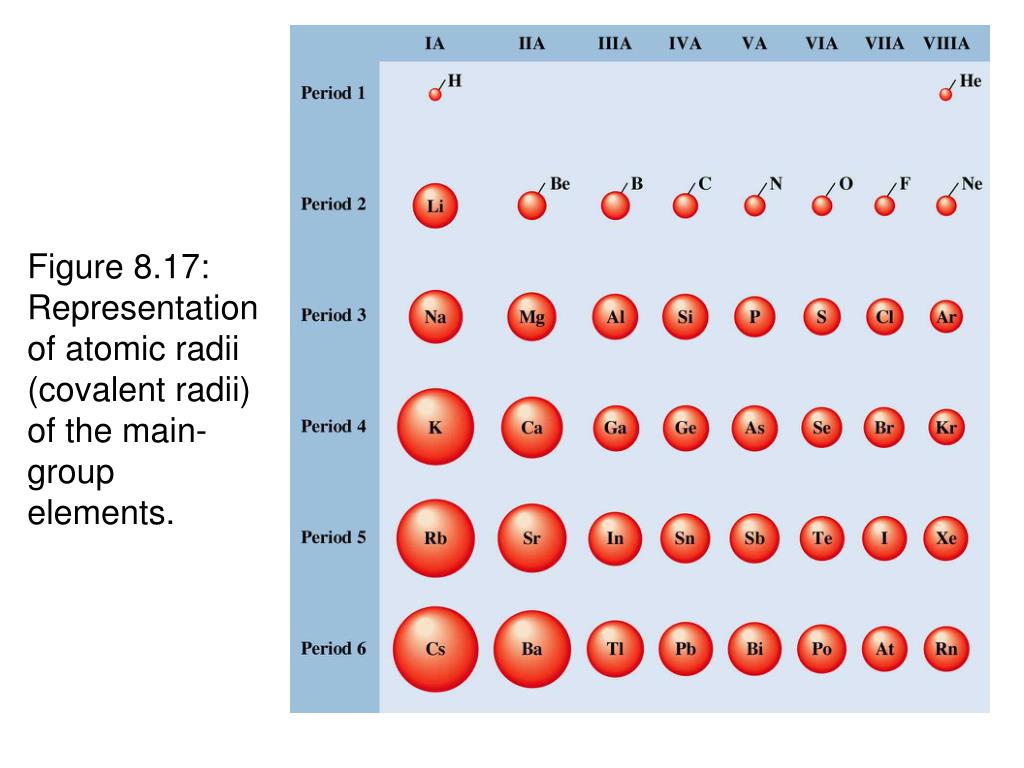
Necessary cookies are absolutely essential for the website to function properly. It sits just below phosphorus on the periodic table, making it a non-metal, and implicating similar atomic radii, the same number of valence electrons, and nearly identical electronegativity and orbital configurations. What is the difference between arsenic and phosphorus?Īrsenic is a chemical analog of phosphorus, just as copper is to iron, mentioned above. O Silicon is a larger stom than phosphorus because phosphorus is in the. ► View Available Hints) O Sicon is a larger atom than phosphorus because phosphones has one more proton than silicon, thereby increasing the attraction for all electrons and decreasing the atomic radius. Neon has a smaller atomic radius than phosphorus because: (A) Unlike neon, phosphorus has electrons present in its third energy level. Which element has a smaller atomic radius than phosphorus? Hence, nitrogen has the smallest atomic radius and antimony has the largest atomic radius. On moving down the group, the distance between the nucleus and outermost shell increases, and the number of new shells increases which increases the atomic radius. Which has the largest atomic radius nitrogen antimony or arsenic? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
